Biochemical Markers of Alzheimer Disease and Dementia - CAM 335HB
Description:
Alzheimer disease (AD) is a neurodegenerative disease defined by a gradual decline in memory, cognitive functions, gross atrophy of the brain, and accumulation of extracellular amyloid plaques and intracellular neurofibrillary tangles (Karch et al., 2014).
Regulatory Status
A search of the FDA Devices database on 07/16/2019 for the terms “peptide”, “Alzheimer”, “tau”, “synuclein”, “chromogranin”, “neuregulin”, “chitinase”, “carnosinase”, “apoE”, and “thread protein” yielded no results pertaining to testing for AD.
On February 15, 2018, the FDA released a statement concerning the advancement of the development of novel treatments for neurological conditions, including Alzheimer’s disease. FDA Commissioner Scott Gottlieb, M.D., states, “Symptoms and progression of neurological diseases can also vary significantly across patients, and even within patients, and across organ systems. Some diseases, like Alzheimer’s, may progress invisibly for years. Once clinical symptoms become apparent, significant function may already be lost. These issues can make drug development more challenging for companies and are deeply frustrating for patients and caregivers living with these serious and life-threatening conditions. The FDA recognizes the urgent need for new medical treatments for many serious conditions including neurological disorders such as muscular dystrophies, amyotrophic lateral sclerosis (ALS), Alzheimer’s disease (AD), migraine and epilepsy. This requires us to become more nimble, collaborative and patient-focused. As part of our ongoing efforts to expand access to safe and effective treatment options across all disease areas and promote innovation, the FDA is modernizing multiple aspects of our drug regulatory programs — including how we communicate scientific and regulatory guidance for drug development" (Gottlieb, 2018). Concurrently, the FDA released a guidance for industry concerning AD for public comment for 90 days. Within the guidance, the FDA states, “FDA supports and endorses the use of diagnostic criteria that are based on a contemporary understanding of the pathophysiology and evaluation of AD. … Important findings applicable to the categorization of AD along its continuum of progression include the presence of pathophysiological changes as measured by biomarkers, the presence or absence of detectable abnormalities on sensitive neuropsychological measures, and the presence or absence of functional impairment manifested as meaningful daily life impact the present with subjective complaints or reliable observer reports" (FDA, 2018). The final draft of the guidance should be released in the future after the public comment period has concluded.
Many labs have developed specific tests that they must validate and perform in house. These laboratory-developed tests (LDTs) are regulated by the Centers for Medicare & Medicaid (CMS) as high-complexity tests under the Clinical Laboratory Improvement Amendments of 1988 (CLIA ’88). As an LDT, the U.S. Food and Drug Administration has not approved or cleared this test; however, FDA clearance or approval is not currently required for clinical use.
Policy:
Application of coverage criteria is dependent upon an individual’s benefit coverage at the time of the request.
- For individuals with Alzheimer disease or mild cognitive impairment, measurement of amyloid beta peptides in cerebrospinal fluid is considered MEDICALLY NECESSARY.
The following does not meet coverage criteria due to a lack of available published scientific literature confirming that the test(s) is/are required and beneficial for the diagnosis and treatment of an individual’s illness.
- Measurement of cerebrospinal fluid biomarkers of Alzheimer disease or dementia not mentioned above (e.g., tau protein, α-synuclein, or neural thread proteins) is considered NOT MEDICALLY NECESSARY.
- Measurement of plasma and/or serum biomarkers of Alzheimer disease or dementia (e.g., tau protein, amyloid beta peptides, neural thread proteins, ApoE, and ApoE4) is considered NOT MEDICALLY NECESSARY.
- Measurement of urinary biomarkers of Alzheimer disease or dementia (e.g., neural thread proteins, amyloid beta peptides, and urinary extracellular vesicle analysis) is considered NOT MEDICALLY NECESSARY.
- The use of multianalyte assays, algorithmic analysis, and/or any other tests not mentioned above for the prognosis, diagnosis, and/or management of Alzheimer disease or dementiais considered NOT MEDICALLY NECESSARY.
Table of Terminology
| Term |
Definition |
| AAN |
American Academy of Neurology |
| AD |
Alzheimer disease |
| AD7c-NTP |
Alzheimer-associated neuronal thread protein |
| ADAD |
Autosomal dominant Alzheimer's disease |
| ALS |
Amyotrophic lateral sclerosis |
| aMCI |
Amnestic mild cognitive impairment |
| APOE |
Apolipoprotein E |
| APOJ |
Apolipoprotein J |
| Aß40 |
Amyloid Beta 40 |
| Aß42 |
Amyloid Beta 42 |
| AUC |
Area under the curve |
| Aβ 25-35 |
Β-amyloid 25-35 |
| CCCDTD |
Canadian Consensus Conference on the Diagnosis and Treatment of Dementia |
| CDC |
Centers For Disease Control and Prevention |
| CLIA ’88 |
Clinical Laboratory Improvement Amendments of 1988 |
| CMS |
Centers for Medicare & Medicaid Services |
| CN |
Cognitively normal |
| CNT |
Carbon nanotubes |
| CSF |
Cerebrospinal fluid |
| CT |
Computerized tomography |
| CU |
Cognitively unimpaired |
| DLB |
Dementia with lewy bodies |
| EFNS |
European Federation of Neurological Societies |
| EV |
Extracellular vesicle |
| FDA |
Food And Drug Administration |
| FDG |
Fluoro-deoxyglucose |
| GBSC |
Global Biomarker Standardization Consortium |
| GSEA |
Gene set analysis |
| HD |
Huntington disease |
| IWG |
International Working Group |
| JPND |
Joint Program — Neurodegenerative Disease Research |
| LDT |
Laboratory-developed test |
| LP |
Lumbar puncture |
| MCI |
Mild cognitive impairment |
| MRI |
Magnetic resonance imaging |
| NFH |
Heavy chain |
| NFL |
Neurofilament light chain |
| NG |
Neurogranin |
| NIA |
National Institute on Aging |
| NICE |
National Institute for Health and Care Excellence |
| NINCDS |
National Institute of Neurological and Communicative Disorders and Stroke |
| NrCAM |
Neuronal cell adhesion molecule |
| NTP |
Neuronal thread protein |
| PD |
Parkinson disease |
| PET |
Positron emission tomography |
| PKCe |
Protein kinase c-epsilon |
| P-tau |
Phosphorylated tau |
| PTP |
Pancreatic thread protein |
| REM |
Rapid eye movement |
| SCD |
Subjective cognitive decline |
| sCJD |
Sporadic Creutzfeldt–Jakob disease |
| SNAP23 |
Synaptosomal-associated protein 23 |
| SORL |
Sortilin-related receptor |
| T-tau |
Total tau |
| USPSTF |
United States Preventive Services Task Force |
Rationale
Alzheimer disease (AD) is a devastating neurodegenerative disease with a strong genetic component and is the predominant form of dementia (60% – 70%). As of March, 2023, more than 55 million people worldwide were living with dementia and it is the seventh leading cause of death and globally is one of the major causes of disability and dependency among older individuals (WHO, 2023). The average lifetime risk of developing AD is 10% – 12%; this risk at least doubles with the presence of a first-degree relative with the disorder (Goldman et al., 2011). The genetic predisposition of AD, even for late-onset AD patients, is estimated to be 60% – 80% (Gatz et al., 2006). According to the Centers for Disease Control and Prevention (CDC), the total adjusted death rates in the U.S. varied according to ethnicity with white, non-Hispanics having a rate of 70.8 per 100,000 individuals as compared to 65.0 and 46.0 per 100,000 for non-Hispanic black and Hispanic individuals (Kramarow & Tejada-Vera, 2019).
Most patients develop clinical symptoms at or after the age of 65 (spontaneous or late-onset AD), however 2% – 10% of patients have an earlier onset of disease (early-onset AD) (Shea et al., 2016). AD is characterized by severe neuronal loss, aggregation of extracellular amyloid β plaques, and intraneuronal tau protein tangles, resulting in progressive deterioration of memory and cognitive functions and ultimately requiring full-time medical care (Sala Frigerio & De Strooper, 2016). There is an enormous burden on public health due to the high costs associated with care and treatment. Aside from drugs that temporarily relieve symptoms, no treatment exists for AD (Van Cauwenberghe et al., 2016).
Many genetic studies have recently identified that late-onset Alzheimer disease is associated with the apolipoprotein E (APOE), apolipoprotein J (APOJ), and sortilin-related receptor (SORL) genes mainly expressed by various types of glial cells such as microglia, oligodendrocytes, and astrocytes; this has helped AD-related research stray from neurons and toward glial cells and neuroinflammation (Arranz & De Strooper, 2019).
The pathological processes of AD and other degenerative dementias are likely well underway before clinical symptoms manifest, therefore, biomarkers may have potential utility in the early diagnosis of dementia (Peterson, 2024). Mild cognitive impairment (MCI) is an intermediate state between normal cognition and dementia, recognizable as an early manifestation of dementia. MCI due to AD is the most common type of MCI (Bennett et al., 2002).
Studies have examined the use of cerebrospinal fluid (CSF) markers for predicting conversion from MCI to dementia. The most replicated CSF biomarkers include tau protein or phosphorylated tau protein and amyloid beta 42 (Aß42) peptide, which may be represented by a low ratio of Aß42 to Aß40 levels, or a low ratio of Aß42 to tau levels. However, these tests vary in sensitivity (36 to 100 percent) and specificity (29 to 91 percent), and in the types of assays used. Recent research notes that the Aß42/40 ratio should be used over the measurement of Aß42 alone, as this ratio gives a more accurate diagnosis when analyzing CSF AD biomarkers (Hansson et al., 2019). Currently, these markers are of marginal clinical utility and do not have an established role in the evaluation of patients in the clinical setting (Peterson, 2024; Wolk & Dickerson, 2024).
Other biomarkers in CSF such as cargo proteins (e.g., chromogranin-B, α-synuclein), carnosinase I, chromogranin A, and NrCAM (neuronal cell adhesion molecule) have been proposed to provide clinical value for assessment of AD. Levels of each of the above CSF proteins are found to be statistically different among clinically defined patient groups with different degrees of cognitive impairment. However, the absence of a clinical treatment makes this relatively invasive test of questionable clinical utility (Schaffer et al., 2015; Wolk & Dickerson, 2024).
Plasma levels of the E4 variant of apolipoprotein E (ApoE4) may be a less invasive option for diagnosing patients. ApoE facilitates the delivery of cholesterol and promotes neuronal functionality and decreased apoE4 levels associated with neuronal degradation are suggestive of AD (Farrer et al., 1997). However, results are inconsistent across various studies. The correlation between altered levels of ApoE and ApoE4 with AD pathology is still not definitive, and standardization of methods is needed (Schaffer et al., 2015).
Studies have been conducted comparing the telomere length of peripheral blood leukocytes with those in the cerebellum (Patel et al., 2011). The shortening of telomere length is indicative of chronic stress on the human body, common in AD patients. However, cerebellar telomere length is not considered a diagnostic tool to evaluate the risk of inherited AD (Patel et al., 2011). Moreover, many other diseases also contain pathologies that induce stress on the body, so results may be confounded with other underlying health problems (Schaffer et al., 2015).
High concentrations of neuronal thread protein (NTP), specifically AD-associated NTP (AD7c-NTP), in urine is found to be representative of AD pathology (Patel et al., 2011). NTP is a brain protein that interacts with antibodies produced against pancreatic thread protein (PTP), a protein that contains structural components highly similar to the fibrils found in neuronal plaques in AD patients (Blennow et al., 2012; Patel et al., 2011). Moreover, AD7c-NTP is reflective of neuronal cell dysfunction. Unfortunately, NTP is more useful in determining the progression of the disease in patients who already have AD and not for early diagnosis (Lonneborg, 2008; Schaffer et al., 2015).
Studies have also identified a potential relationship between nanoscale extracellular vesicles (exosomes) and AD. Researchers note that exosomes may be an important factor in the progression of AD pathogenesis, but first need to identify the underlying AD-related mechanisms (Jiang et al., 2019).
Other media, such as saliva, have been proposed to provide diagnostic information for AD. A total of 6,230 metabolites from saliva were tested, and three were found to differentiate between MCI, AD, and cognitively normal patients (Huan et al., 2018).
None of these tests or biomarkers are valid as a stand-alone diagnostic test. The lack of standardized techniques makes diagnostic accuracy across all scenarios difficult to achieve. Current AD diagnostic standards using evaluation of clinical presentation have maintained a high level of accuracy, combined with the lack of a clinical treatment make all early AD diagnostic tests and biomarkers of limited clinical utility (François et al., 2019; Schaffer et al., 2015). However, research criteria have incorporated both molecular and topographic biomarker data into the research definitions of both symptomatic and pre-symptomatic forms of AD, anticipating that once biomarkers become more standardized, they will be incorporated into clinical diagnostic algorithms for AD (Morris et al., 2014; Wolk & Dickerson, 2024).
Proprietary Testing
Proprietary tests exist for assessment of AD biomarkers. C2N Diagnostics offers PrecivityAD™, a blood test that measures the ratio of Aß42 to Aß40 and ApoE detection. C2N Diagnostics received a “Breakthrough Device Designation” from the FDA in January 2019 for their test measuring the ratio of Aß42 to Aß40 (C2N, 2019). Fujirebio Diagnostics offers the in vitro Lumipulse® G β-Amyloid Ratio (1-42/1-40) test, which combines the results of the Lumipulse® G β-Amyloid 1-42 and Lumipulse® G β-Amyloid 1-40 to create a ratio of beta-amyloid 1-42 and beta-amyloid 1-40 concentrations in CSF with the LUMIPULSE G1200 system (Fujirebio, 2022). This is intended to predict the likelihood of amyloid plaque formation in potential AD. This assay received the “Breakthrough Device Designation” from the FDA in May 2022 (FDA, 2022).
Roche Diagnostics received 501(k) clearance from the FDA in 2022 for their Elecsys® beta-Amyloid (1-42) CSF II (Abeta42) and Elecsys® Phospho-Tau (181P) CSF (pTau181) assays in 2022 for adults 55 years and older who are evaluated for the disease and other cognitive impairments to generate a pTau181/Abeta42 ratio value. The company cites that these assays “achieve 90% concordance with the Amyloid PET scan imaging and have the potential to provide a more affordable and accessible routine option to confirm the presence of amyloid in the brain.” They can also detect pathology in earlier stages of disease due to the correlative changes in biomarkers (Roche, 2022). In June 2023, Roche Diagnostics also received 501(k) clearance from the FDA for the Elecsys® beta-Amyloid (1-42) CSF II (Abeta42) and Elecsys® Total-Tau CSF assays (tTau) in the same population through the tTau/Abeta42 ratio, and will be available in Q4 2023. The company endorses that these assays provide a cost-effective, more widely available alternative to the recommended PET imaging option with minimal radiation exposure. The ratio would be “consistent with a negative amyloid PET scan if the result is less than or equal to the cutoff (negative), and with a positive amyloid PET scan if the result is above the ratio cutoff (positive) (Roche, 2023).
On July 6, 2023, Quanterix® launched the LucentAD test, which measures serum levels of tau protein phosphorylated at Thr181 (p-Tau 181), which is a marker of AD pathology. It is intended to assist in the diagnostic evaluation of AD with other tools, but clues providers into a patient’s likelihood of amyloid-related pathology. It is not currently approved by the FDA, but has been studied in conjunction with the drug lecanemab in its effectiveness for treating AD therapy response (BusinessWire, 2023).
Lecanemab is an antibody intravenous (IV) infusion therapy. It works by targeting and removing beta-amyloid from the brain. It has received traditional approval from the U.S. Food and Drug Administration (FDA) to “treat early Alzheimer's disease, including people living with mild cognitive impairment (MCI) or mild dementia due to Alzheimer's disease who have confirmation of elevated beta-amyloid in the brain” (Alzheimer's Association, 2024). Lecanemab lowers beta-amyloid in the brain, reducing cognitive and functional decline in people living with early Alzheimer's. This treatment is appropriate for people with early Alzheimer's who have been confirmed to have elevated beta-amyloid levels. The test is appropriate in those with elevated beta-amyloid levels and as such, before treatment could be considered, the physician would first need to confirm the presence of beta-amyloid plaques. Currently, the FDA does not specify a diagnostic tool to determine elevated beta-amyloid. However, some examples of tools to diagnose beta-amyloid elevation include PET scan or lumbar puncture (CSF) tests (Alzheimer's Association, 2024).
The Syn-One Test offered by CND Life Sciences is intended to aid in the diagnosis of synucleinopathies, which includes Parkinson Disease (PD), dementia with Lewy bodies, multiple system atrophy, and pure autonomic failure. Using a skin biopsy, the test detects (through immunostaining) abnormal (phosphorylated) alpha-synuclein (P-Syn) in skin nerve fibers. The Syn-One pathology report also includes results for small fiber neuropathy and amyloidosis (CND Life Sciences).
There is a growing body of evidence confirming the association of P-Syn with various synucleinopathies and P-Syn’s potential utility as a diagnostic biomarker. Donadio et al. (2019) reported “excellent” inter- and intra-laboratory reproducibility when using the skin biopsy technique to assay for P-Syn, providing increased confidence that this approach may be feasible across institutions. Levine et al. (2021) showed that P-Syn might be useful as a marker for other disease states (postural orthostatic tachycardia syndrome), expanding the potential applications of the analyte for different clinical scenarios.
Despite the growing support, other studies have highlighted some concerns: Kim et al. (2019) reviewed the available data regarding the use of cutaneous alpha-synuclein as a marker for synucleinopathies and confirmed that a high specificity is consistently reported across studies, but that sensitivity measurements can vary widely. The authors suggest that this is likely influenced by the specific type of disease, as well as differences in design and methodology (i.e., biopsy site, tissue thickness, or fixation methods). Waqar et al. (2023) published a similar review that emphasized the variability in reported sensitivity of P-Syn detection from skin biopsies, as well as the small sample sizes in many of the published studies to date. The authors do acknowledge, however, the many advantages of using skin samples, including the low cost and high patient tolerance of the technique.
Clinical Utility and Validity
Dage et al. (2016) studied the correlation of tau protein levels (in plasma) with neuronal damage. A total of 378 cognitively normal (CN) patients were examined, along with 161 patients with mild cognitive impairment (MCI). Baseline plasma tau protein levels were measured. The authors found that plasma tau levels were higher in MCI patients compared to CN patients (4.34 pg/mL for MCI compared to 41.4 pg/ML for CN, p = .078). The authors also performed a regression accounting for age, gender, education, and ApoE, which suggested that higher plasma tau levels were associated with worse memory loss and abnormal cortical thickness (Dage et al., 2016).
Lewczuk et al. (2017) compared the ratio of Aβ42/40 to just Aβ42 as measurements of clinical AD. A total of 200 patients (150 PET-negative, 50 PET-positive for amyloid) were examined and compared to the positron emission tomography (PET) results. The authors found that the ratio of Aβ42/40 agreed more strongly with the PET results (89.4% concordance compared to 74.9% concordance for Aβ42 only). A larger area under the curve was found for the Aβ42/40 measurement compared to just Aβ42 (0.936 compared to 0.814). The authors concluded that “the CSF Aβ42/40 ratio is superior to Aβ42 alone as a marker of amyloid-positivity by PET” (Lewczuk et al., 2017).
Talwar et al. (2016) performed a meta-analysis on CSF ApoE levels in AD patients. Twenty-four studies, including 1064 AD cases and 1338 healthy controls, were reviewed. The authors found that although the total sample did not indicate a significant association between AD and ApoE levels, a subgroup analysis controlling for sample size (n > 43) indicated significantly lower ApoE levels in AD patients compared to controls. The authors considered CSF ApoE levels to have “potential” as an indicator of AD association (Talwar et al., 2016).
Wang et al. (2018) evaluated the clinical value of α-synuclein in MCI and AD. The investigators added α-synuclein and phosphorylated α-synuclein to a biomarker panel containing Aβ42, tau, and phosphorylated tau and evaluated the new panel’s performance. A total of 729 CSF samples were taken. The phosphorylated version of α-synuclein was found to weakly associate with diagnosis at baseline, but total α-synuclein was not. CSF α-synuclein was found to predict the Alzheimer Disease Assessment Scale-Cognitive, memory, executive function, and progression from MCI to AD. Longitudinal biomarker changes were not found to differ between groups. Overall, α-synuclein was found to potentially better predict AD changes better than the classic biomarkers (Wang et al., 2018).
Zhang et al. (2014) performed a meta-analysis focusing on urinary Alzheimer-associated neuronal thread protein (AD7c-NTP)’s diagnostic ability for AD. Nine studies were reviewed for probable and possible AD, and the authors evaluated AD7c-NTP’s sensitivity at 0.87, specificity at 0.89, positive likelihood ratio at 8.13, and negative likelihood ratio at 0.15 (Zhang et al., 2014).
Wang et al. (2019) explored the potential of urinary extracellular vesicle (EV) biomarkers in neurological disorders, including AD, Parkinson Disease (PD), and Huntington Disease (HD). A discovery cohort of 50 individuals was used to create the initial set of EV proteins and a set of 108 individuals was used to further develop the list of biomarkers. The authors identified “hundreds” of commonly expressed EV proteins with stable expression. SNAP23 and calbindin were most elevated in PD cases, with an 86% prediction of diagnostic success in the discovery cohort and 76% prediction of diagnostic success in the replication cohort. Moreover, “Broad Gene set analysis (GSEA) further reveals a prominent link to Alzheimer disease with 10.4% of the genes known to be down-regulated in the brains from patients with Alzheimer disease identified in urinary EVs” (Wang et al., 2019).
Liu et al. (2018) examined the urinary metabolic profile of β-amyloid 25-35 (Aβ 25-35)-injected rats. This was intended to establish AD in the rats, allowing the impairment of spatial learning and memory to be tested in the rats after eight weeks. The authors identified the characteristic AD symptoms after eight weeks (cognitive dysfunction, hippocampus damage, Aβ formation and tau phosphorylation) as well as 45 altered metabolites involving eight metabolic pathways. The investigators concluded that “pathogenesis of AD was mainly due to gut microbiome dysbiosis, inhibition of energy metabolism, oxidative stress injury and loss of neuronal protective substances” (Liu et al., 2018).
Fossati et al. (2019) studied the correlation of plasma tau with cerebrospinal fluid (CSF) tau and phosphorylated tau (P-tau). A total of 97 subjects were included (68 healthy controls and 29 AD patients). Plasma tau was found to be higher in AD patients compared to healthy controls (area under curve: 0.79). However, CSF tau and plasma tau were “poorly” correlated. The addition of plasma tau to the receiver operating curve of CSF tau increased the area under curve to 0.82 from 0.80 and increased the curve of P-tau to 0.88 from 0.87. The authors concluded that “adding plasma tau to CSF tau or P-tau improves diagnostic accuracy, suggesting that plasma tau may represent a useful biomarker for AD” (Fossati et al., 2019).
Tatebe et al. (2017) developed an immunoassay to quantify plasma p-tau181. Three cohorts were used to validate the assay. In the first cohort (20 AD patients, 15 controls), the tau levels were found to be higher in the AD patients (0.171 ± 0.166 pg/ml in AD versus 0.0405 ± 0.0756 pg/ml in controls). In the second cohort (20 Down Syndrome patients, 22 controls), the tau levels were higher in the Down Syndrome patients (0.767 ± 1.26 pg/ml in DS versus 0.0415 ± 0.0710 pg/ml in controls). Finally, in the third cohort (eight AD patients, three other neurological diseases), the tau levels were found to correlate well with the CSF tau levels (r2 = 0.4525). Overall, the authors suggested that “that the plasma p-tau181 is a promising blood biomarker for brain AD pathology” (Tatebe et al., 2017).
Shen et al. (2019) completed a meta-analysis review of 170 studies to research the role of inflammatory markers in AD and MCI. Increased periphery levels, compared to controls, were found with many types of biomarkers including high-sensitivity C reactive protein, p < 0.05; interleukin-6, p < 0.005; soluble tumour necrosis factor receptor 1, p < 0.005; soluble tumour necrosis factor receptor 2, p < 0.005; alpha1-antichymotrypsin, p < 0.005; IL-1β, p < 0.05; soluble CD40 ligand, p < 0.05; CSF levels of IL-10, p < 0.05; monocyte chemoattractant protein-1, p < 0.005; transforming growth factor-beta 1, p < 0.05; soluble triggering receptor expressed on myeloid cells2, p < 0.001; YKL-40, p < 0.001; α1-ACT, p < 0.001; nerve growth factor, p < 0.005; and visinin-like protein-1, p < 0.005 (Shen et al., 2019). The authors conclude that all the significant relationships found in this large meta-analysis help to support “the notion that AD and MCI are accompanied by inflammatory responses in both the periphery and CSF” (Shen et al., 2019).
Palmqvist et al. (2019) analyzed two different, cross-sectional, multicenter studies (n = 1079). The CSF Aβ42/Aβ40 ratio was used to identify AD via Elecsys immunoassays from Roche Diagnostics; further, plasma neurofilament light chain (NFL), heavy chain (NFH), and APOE genotype were also analyzed in the first cohort of patients (n = 842). “In cohort 1, plasma Aβ42 and Aβ40 predicted Aβ status with an area under the receiver operating characteristic curve (AUC) of 0.80 (95% CI, 0.77 – 0.83). When adding APOE, the AUC increased significantly to 0.85 (95% CI, 0.82 – 0.88)” (Palmqvist et al., 2019). Cohort 2 had similar results with a slightly higher AUC (0.86; 95% CI, 0.81 – 0.91). The authors conclude by stating that “Plasma Aβ42 and Aβ40 measured using Elecsys immunoassays predict Aβ status in all stages of AD with similar accuracy in a validation cohort. Their accuracy can be further increased by analyzing APOE genotype” (Palmqvist et al., 2019).
Kim et al. (2020) studied the diagnostic utility of multiplexed sensing to detect multiple AD biomarkers (t-tau, p-tau181, Aβ42, and Aβ40) in human plasma using densely aligned carbon nanotubes (CNT). The CNT sensor assay exhibited superior sensitivity and precision, enabling the platform to accurately quantify AD biomarkers despite the hundreds of other agents in the blood plasma. The densely aligned CNT sensor array was 10 – 103 times more sensitive than the commercially available sandwich-type or enzyme-linked immunosorbent assay. The authors conclude that "by measuring the levels of t-tau/Aβ42, p-tau181/Aβ42, and Aβ42/Aβ40 in clinical blood samples, the sensor array successfully discriminates the clinically diagnosed AD patients from healthy controls with an average sensitivity of 90.0%, a selectivity of 90.0%, and an average accuracy of 88.6%" (Kim et al., 2020).
Simrén et al. (2021) studied the diagnostic and prognostic potential of plasma biomarkers in Alzheimer disease. Various biomarkers, including phosphorylated-tau181 (P-tau181), neurofilament light, amyloid-β (Aβ42/40), total-tau and glial fibrillary acidic protein, were analyzed in 99 cognitively unimpaired (CU) patients, 107 mild cognitive impairment (MCI) patients, and 103 Alzheimer disease (AD) patients. According to the results, P-tau181 significantly outperformed all biomarkers in differentiating AD dementia from CU. Higher P-tau181 value was associated with increased cognitive decline and gray matter loss in temporal regions. The authors conclude that "these findings highlight the potential value of plasma P-tau181 as a non-invasive and cost-effective diagnostic and prognostic biomarker in AD" (Simrén et al., 2021).
Qu et al. (2021) performed a systematic review and meta-analysis of 150 studies aiming to evaluate the effect of AD biomarkers on blood. The authors performed a “random-effects meta-analysis based on the ratio of means method and multivariable-adjusted effect estimates.” The results demonstrated that T-tau, P-tau and Nfl increased, and that AβPPR decreased from controls to amnestic MCI (aMIC) to AD. Aβ42, Aβ42/40, and P-tau217 all had valid diagnostic accuracy. The authors conclude that the significant changes in core blood biomarkers support that “biomarkers were strongly valid in identifying AD” (Qu et al., 2021).
Chen et al. (2021) performed a meta-analysis of 17 studies aimed at calculating the diagnostic accuracy of blood-based biomarkers. The authors compared the diagnostic odds ratio (DOR) of biomarkers between controls, AD, and aMCI conditions. When comparing AD and control groups, the plasma Aβ42 DOR was 32.2 (sensitivity = 88 %, specificity = 81 %), the plasma Aβ oligomer DOR was 29.1 (sensitivity = 80 %, specificity = 88 %), and the plasma tau DOR was 52.1 (sensitivity = 90 %, specificity = 87 %). When comparing aMCI and controls, the plasma Aβ42 DOR was 60.4 (sensitivity = 86 %, specificity = 90 %), and the plasma tau DOR was 49.1 (sensitivity = 79 %, specificity = 94 %). The authors conclude that blood-based biomarkers are “minimally invasive and cost-effective tools for detecting AD; however, the evidence for detecting aMCI was still limited” (Chen et al., 2021).
Yoong et al. (2021) performed a systematic review and meta-analysis of 13 studies aiming to address the prognostic utility of a new CSF biomarker: Neurogranin (Ng). Core CSF biomarkers such as Aβ42, T-tau, and P-tau can support AD diagnosis, but cannot predict AD progression. Ng has been shown to predict cognitive decline. The authors found evidence that CSF Ng can predict Mini-Mental State Examination (MMSE) decline in Aβ+ MCI patients and the decline of memory and executive function in MCI. Additionally, CSF Ng/Aβ42 was also found likely to predict cognitive decline. The authors conclude that CSF Ng may be an applicable AD biomarker, but more studies are required to validate 548309its use (Yoong et al., 2021).
Nojima et al. (2022) investigated the clinical utility of measuring CSF biomarkers through the LUMIPULSE® system in correlation with Aβ deposition status confirmed by amyloid PET. From 199 CSF samples from patients with confirmed AD and underwent amyloid PET, measurements of Aβ 1–40 (Aβ40), Aβ 1–42 (Aβ42), total tau (t‐Tau), and phosphorylated tau‐181 (p‐Tau181) using the LUMIPULSE system were taken and analyzed with a multivariable logistic regression model. Through this, they were able to determine that there was diagnostic agreement between the biomarker levels and amyloid PET imaging, and that there was statistical significance in the association between amyloid PET status and Aβ40 and Aβ42, with the ratios providing better diagnostic agreement than single biomarkers alone. Researchers also determined that the statistically significant correlation between the Aβ42/Aβ40 ratio and p‐Tau181 may render a plausible utility in predicting brain Aβ pathology. The CSF findings may also potentially draw parallels to benefits with measuring blood plasma levels of the AD biomarkers with high-sensitivity assays, but the “plasma biomarker levels could be affected by small measurement variations caused by preanalytical handling and analytical performance, leading to misclassification (i.e., false‐negative or false‐positive for Aβ pathology)”(Nojima et al., 2022).
National Institute of Neurological and Communicative Disorders and Stroke (NINCDS) and the Alzheimer’s Disease and Related Disorders Association (ADRDA)
In 1984, the NINCDS and ADRDA developed clinical criteria for the diagnosis of AD. While evidence to date has used NINCDS/ADRDA’s AD classification, in 2011, the National Institute on Aging and the Alzheimer’s Association workgroup revised diagnostic criteria for diagnosis of dementia due to Alzheimer’s disease (McKhann et al., 2011).
The biomarkers reviewed in this policy are included in a category among revisions to AD diagnostic criteria — “probable AD dementia with evidence of the AD pathophysiological process." However, the diagnostic criteria workgroup publication noted “we do not advocate the use of AD biomarker tests for routine diagnostic purposes at the present time. There are several reasons for this limitation: 1) the core clinical criteria provide very good diagnostic accuracy and utility in most patients; 2) more research needs to be done to ensure that criteria that include the use of biomarkers have been appropriately designed, 3) there is limited standardization of biomarkers from one locale to another, and 4) access to biomarkers is limited to varying degrees in community settings. Presently, the use of biomarkers to enhance certainty of AD pathophysiological process may be useful in three circumstances: investigational studies, clinical trials, and as optional clinical tools for use where available and when deemed appropriate by the clinician” (McKhann et al., 2011).
Alzheimer’s Association
The Alzheimer’s Association has initiated a quality control program for CSF markers, noting that “Measurements of CSF AD biomarkers show large between laboratory variability, likely caused by factors related to analytical procedures and the analytical kits. Standardization of laboratory procedures and efforts by kit vendors to increase kit performance might lower variability, and will likely increase the usefulness of CSF AD biomarkers” (Mattsson et al., 2011).
In 2013, the Alzheimer’s Association published recommendations for operationalizing the detection of cognitive impairment in the primary care setting (Cordell et al., 2013). It stated that “the use of biomarkers (e.g., CSF tau and beta amyloid proteins, amyloid tracer positron emission tomography scans) was not considered as these measures are not currently approved or widely available for clinical use.”
In 2018, a multidisciplinary group of the Alzheimer’s Association published criteria for lumbar puncture and CSF testing in the diagnosis of AD. The committee recommends CSF biomarker testing for six clinical indications deemed appropriate, as listed in the table below.
Table 1: Clinical indications for appropriate use of LP and cerebrospinal fluid testing in the diagnosis of AD (Shaw et al., 2018)
| No. |
Indication |
Ratings |
| 1 |
Cognitively unimpaired and within normal range functioning for age as established by objective testing; no conditions suggesting high risk and no SCD [subjective cognitive decline] or expressed concern about developing AD |
Inappropriate |
| 2 |
Cognitively unimpaired patient based on objective testing, but considered by patient, family informant, and/or clinician to be at risk for AD based on family history |
Inappropriate |
| 3 |
Patients with SCD (cognitively unimpaired based on objective testing) who are considered to be at increased risk for AD |
Appropriate |
| 4 |
Patients with SCD (cognitively unimpaired based on objective testing) who are not considered to be at increased risk for AD |
Inappropriate |
| 5 |
MCI that is persistent, progressing, and unexplained |
Appropriate |
| 6 |
Patients with symptoms that suggest possible AD |
Appropriate |
| 7 |
MCI or dementia with an onset at an early age ( < 65) |
Appropriate |
| 8 |
Meeting core clinical criteria for probable AD with typical age of onset |
Appropriate |
| 9 |
Symptoms of REM sleep behavior disorder |
Inappropriate |
| 10 |
Patients whose dominant symptom is a change in behavior (e.g., Capgras Syndrome, paranoid delusions, unexplained delirium, combative symptoms, and depression) and where AD diagnosis is being considered |
Appropriate |
| 11 |
Use to determine disease severity in patients having already received a diagnosis of AD |
Inappropriate |
| 12 |
Individuals who are apolipoprotein E (APOE) ɛ4 carriers with no cognitive impairment |
Inappropriate |
| 13 |
Use of LP in lieu of genotyping for suspected ADAD mutation carriers |
Inappropriate |
| 14 |
ADAD mutation carriers, with or without symptoms |
Inappropriate |
Abbreviations: AD, Alzheimer's disease; LP, lumbar puncture; REM, rapid eye movement; SCD, subjective cognitive decline; ADAD, autosomal dominant Alzheimer's disease; MCI, mild cognitive impairment.
The workgroup has also identified several gray areas where more research is needed. The authors note that “One question that will need further data is whether measuring a ratio of CSF Aβ42/40 yields better diagnostic performance than measuring Aβ42 alone. Another question is how to characterize neurodegeneration using CSF biomarkers, and whether neurodegeneration in the absence of positive amyloid biomarkers predicts progression in persons with MCI” (Shaw et al., 2018). Further, the authors also state that “much more work is needed to document the potential impact of CSF AD biomarker testing on clinical outcomes in patients across the spectrum of AD” (Shaw et al., 2018).
In 2022, the Alzheimer’s Association published a recommendation on the appropriate use of blood-based biomarkers (BBMs) in AD. In this recommendation, they note that BBMs are showing promise in revolutionizing the diagnostic and prognostic work-up of AD. However, they still caution that BBMs should not yet be used as primary endpoints in pivotal trials. The recommend that BBMS may be cautiously used in specialized memory clinics, but additional data are needed before BBMS can be used as a stand-alone diagnostic AD marker or before they should be considered for use in primary care. While they do recognize that BBMs are useful as (pre)-screeners for clinical trials, they only cautiously recommend the use of BBMs outside the clinical trial setting:
“Recommendations of the use of AD-associated BBMs in clinical trials and practice:
Use of BBMs in specialized memory clinic settings:
(5) BBMs (with established thresholds) should currently only be used in symptomatic patients at specialist clinics and the results should be confirmed whenever possible with CSF or PET. Additional data are needed before use of BBMs as stand-alone diagnostic markers.
Use of BBMs in primary care:
(6) Additional data are needed for use of BBMs in primary care” (Hansson et al., 2022).
“That said, the implementation of such markers in trials and practice must be done in a careful and controlled way not to accidentally cause more harm than good. Much more research is therefore needed before widespread clinical use of BBMs as we have outlined above. Such research is also needed before the community can establish Appropriate Use Criteria for clinical use of BBMs, which is a prerequisite for general use of such markers in the clinic. However, the acquired experience from implementation of CSF AD biomarkers and Aβ-PET in many countries will ensure rapid validation of relevant BBMs in the first contexts of use, including trials and specialized memory clinics. The implementation of BBMs in primary care will likely take much longer, because relevant and high-quality research studies on AD-related BBMs in this setting are very few, but hopefully more prospective studies will be launched in the coming years using relevant and accurate reference standards” (Hansson et al., 2022)
Expert Working Group for the EU Joint Program — Neurodegenerative Disease Research (JPND) BIOMARKAPD Program
An expert working group, comprised of 28 international members, was convened to develop recommendations for CSF AD biomarkers in the diagnostic evaluation of dementia. “The working group recommended using the CSF biomarkers in MCI as an add‐on to clinical evaluation alone for predicting functional decline or progression to AD dementia and, based on the available evidence, the recommendation was strong. However, in comparison with the outcome of using hippocampal atrophy as a biomarker, the working group issued a weak recommendation to incorporate CSF biomarkers in the diagnostic workup compared with hippocampal atrophy. Because of insufficient evidence, the working group could not recommend CSF biomarkers as an alternative to FDG‐PET or amyloid‐PET in predicting future decline or conversion. The working group recommended using CSF biomarkers to inform future disease management, but the strength of this recommendation was weak because of the small amount of evidence” (Simonsen et al., 2017).
Six clinical questions were asked by Simonsen et al. (2017):
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers identify or exclude AD as the cause of MCI?”
- Final recommendation: N/A
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers predict conversion to AD dementia within 3 years?”
- Final recommendation: Yes, strong
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers predict functional or cognitive decline?”
- Final recommendation: Yes, strong
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers change disease management?”
- Yes, weak
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers improve patient well‐being?”
- Yes, weak
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers reduce health care costs?”
- No, weak
Additional recommendations were made by Herukka et al. (2017) for CSF AD biomarkers in the diagnostic evaluation of mild cognitive impairment. The same six clinical questions were asked as above by Simonsen et al.:
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers identify or exclude AD as the cause of MCI?”
- Final recommendation: N/A
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers predict conversion to AD dementia within 3 years?”
- Final recommendation: Yes, strong
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers predict functional or cognitive decline?”
- Final recommendation: Yes, strong
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers change disease management?”
- Yes, weak
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers improve patient well‐being?”
- Yes, weak
- “In patients with MCI, will CSF biomarkers (alone or in combination) compared with (A) clinical measures alone and/or (B) other imaging biomarkers reduce health care costs?”
- No, weak
National Institute on Aging (NIA, NIH) and Alzheimer’s Association
In 2011, the National Institute on Aging and Alzheimer’s Association workgroups published guidelines for the diagnosis of AD. The authors also note that “Two fundamental issues about individuals with MCI may be answered by the use of biomarkers: (1) To establish support for the underlying etiology of the clinical syndrome in an individual with MCI, which will have major importance for choosing the correct therapy, when effective treatments are available. (2) To determine the likelihood of cognitive and functional progression for an individual MCI patient to a more severe stage of MCI or to dementia, and the likelihood that this progression will occur within a defined period” (Albert et al., 2011). The authors also note that “in these recommendations, CSF tau is considered to be a strong marker of the neuronal injury associated with AD. However, the two biomarkers in combination are extremely informative. Together with low CSF Aβ42, elevated CSF tau provides a high likelihood of progression to AD in patients with MCI;” however, because many biochemical events may be associated with AD, the authors confirm that “Additional work in this area is needed to know how useful these markers will be” (Albert et al., 2011).
In 2018, guidelines were published by the National Institute on Aging and Alzheimer’s Association for the preclinical, mild cognitive impairment, and dementia stages of AD, and are intended for use in observational and interventional research, not routine clinical care. These guidelines state that “there is now a growing consensus that application of biomarkers should be harmonized conceptually across the disease continuum and that biomarkers of neurodegeneration are not equivalent to those reflecting amyloid and pathologic tau accumulation” (Jack et al., 2018). Further, regarding the guidelines noted above from 2011, the authors state that “Studies published since 2011 have reinforced the idea that certain imaging and CSF biomarkers are valid proxies for neuropathologic changes of AD … additional research has highlighted the fact that measures of neurodegeneration or neuronal injury that are commonly used in AD research — magnetic resonance imaging (MRI), fluoro-deoxyglucose (FDG) PET, and CSF total tau (T-tau) — are not specific for AD but rather are nonspecific indicators of damage that may derive from a variety of etiologies, for example, cerebrovascular injury” (Jack et al., 2018). The authors also state that the “data firmly establish that more advanced disease defined by biomarkers predicts greater likelihood of and more rapid cognitive decline. Thus, a solid evidence base exists proving that combinations of biomarker abnormalities are useful for staging the Alzheimer’s continuum” (Jack et al., 2018).
Global Biomarker Standardization Consortium (GBSC)
The GBSC of the Alzheimer’s Association has noted that before biomarkers can be used in clinical practice, they “must be standardized and validated on a global scale” (GBSC, 2024).
American Academy of Neurology (AAN)
In 2018, a guideline was issued as an update to the 2001 AAN guideline on mild cognitive impairment (MCI) and endorsed by the Alzheimer’s Association. This guideline was reaffirmed in 2021 (AAN, 2021). The panel determined that the field of biomarkers is rapidly evolving. And, according to the panel, there are no biomarkers that that could clearly predict progression in patients with MCI. They have provided the following recommendations:
Recommendation A7a
For patients and families asking about biomarkers in MCI, clinicians should counsel that there are no accepted biomarkers available at this time (Level B).”
Recommendation A7b
For interested patients, clinicians may discuss the option of biomarker research or refer patients or both, if feasible, to centers or organizations that can connect patients to this research (e.g., subspecialty centers, Trial Match, ClinicalTrials.gov) (Level C).”
In 2001, the Quality Standards Committee of the American Academy of Neurology issued a “Practice parameter: Diagnosis of dementia (an evidence-based review).” Relevant statements to the current policy include the following:
"... no laboratory tests have yet emerged that are appropriate or routine use in the clinical evaluation of patients with suspected AD. Several promising avenues genotyping, imaging and biomarkers are being pursued, but proof that a laboratory test has value is arduous. Ultimately, the putative diagnostic test must be administered to a representative sample of patients with dementia who eventually have pathologic confirmation of their diagnoses. A valuable test will be one that increases diagnostic accuracy over and above a competent clinical diagnosis."
"There are no CSF or other biomarkers recommended for routine use in determining the diagnosis of AD at this time” (Knopman et al., 2001)
Dementia With Lewy Bodies (DLB) Consortium
The DLB Consortium published a consensus report on the diagnosis and management of dementia with Lewy bodies, which are characteristic of Alzheimer’s Disease and other neurological conditions. The Consortium states that “direct biomarker evidence of LB-related pathology is not yet available for clinical diagnosis” (McKeith et al., 2017).
Consensus of the Task Force on Biological Markers in Psychiatry of the World Federation of Societies of Biological Psychiatry
The Federation published an update on cerebrospinal fluid (CSF) and blood biomarkers for neurodegenerative dementias. The Federation considers blood-based biomarkers to “offer an ideal complementary step to advanced CSF and neuroimaging biomarkers and can serve as the first-step in a multi-stage process”, although these biomarkers still require validation and “a great deal of additional work” (Lewczuk et al., 2018).
International Working Group (IWG)
Dubois et al. (2014) published a position paper which presents a new diagnostic algorithm for AD which states: “Aβ1–42 and tau (T-tau or P-tau) should be used in combination, and the CSF AD signature, which combines low Aβ1 and high T-tau or P-tau concentrations, significantly increases the accuracy of AD diagnosis even at a prodromal stage. This combination reaches a sensitivity of 90% – 95% and a specificity of about 90% in AD. CSF biomarkers cannot be used as standalone tests and should be interpreted in a larger clinical context with confounding factors considered. An important concern is the large variability in CSF measures between laboratories and across techniques, and the lack of agreement on cutoff thresholds. These variations have made direct comparison of study results difficult. Several programmes of standardisation, including the Alzheimer’s Association Quality Control programme for CSF biomarkers, initiatives within the Joint Program for Neurodegenerative Diseases, and the Global Biomarker Standardisation Consortium, and by industry, will minimise between-laboratory variations in the future and allow identification of uniform cutoff levels.” In their 2021 IWG position paper, the group states “Overall, evidence for the use of biomarkers in clinical practice remains highly disputed and suffers from a dearth of evidence-based data to recommend biomarker assessments for cognitively unimpaired individuals.” (Dubois et al., 2021).
The IWG describes specific biochemical evidence in their definitions of AD:
“In-vivo evidence of Alzheimer’s pathology (one of the following):
- Decreased Aβ1–42 together with increased T-tau or P-tau in CSF
- Increased tracer retention on amyloid PET
- AD autosomal dominant mutation present (in PSEN1, PSEN2, or APP)” (Dubois et al., 2014).
In their updated proposed recommendations, the IWG included the following relevant information:
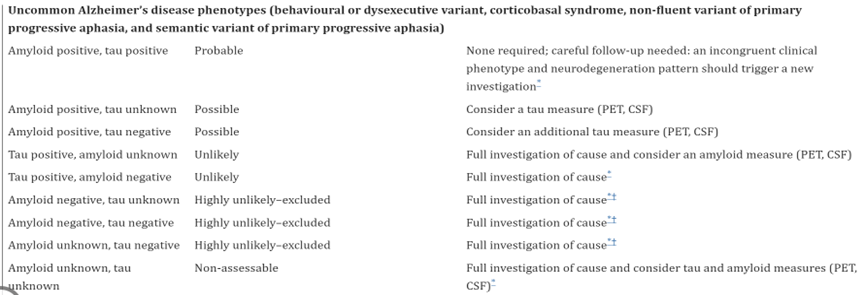
- “The diagnosis of Alzheimer’s disease is clinical-biological. It requires the presence of both a specific clinical phenotype of Alzheimer’s disease (phenotype positive) and biomarker evidence of Alzheimer’s disease pathology (amyloid-positive and tau positive).
- In people who have … common phenotypes, amyloid and tau biomarker positivity establishes an Alzheimer’s disease diagnosis (table 2). The positivity of both amyloid and tau biomarkers is required because an amnestic phenotype with only amyloid positivity is not specific to Alzheimer’s disease and is seen in other neurodegenerative diseases with amyloid copathology (including LATE and dementia with Lewy bodies) or in patients with cerebral amyloid angiopathy and amnestic vascular cognitive impairment. However, an isolated amnestic syndrome of the hippocampal type with only tau biomarker positivity can occur in primary age-related tauopathy or in atypical presentations of mixed 3 repeat or 4 repeat tau frontotemporal lobar degeneration. Finally, uncommon phenotypes with positive Alzheimer’s disease biomarkers should not be a-priori classified as an established Alzheimer’s disease (table 2); in such cases the clinician could deem that Alzheimer’s disease is not the dominant pathology driving the clinical phenotype but only a copathology.
- Recommended biomarker measures for amyloid β pathology are low CSF Aβ42, increased CSF Aβ40–Aβ42 ratio (which is, if possible, preferred to low CSF Aβ42) or high tracer retention in amyloid PET. For tau pathology, we recommend high CSF phosphorylated tau (not total tau because of low specificity) or increased ligand retention in tau PET. Recommendation of amyloid PET and tau PET for use in clinical practice is conditional on regulatory approval and reimbursement by payers in different countries.
- CSF investigation is prioritized because it provides simultaneous information on the two types of biomarkers (amyloid β and tau) and is less expensive than amyloid PET, tau PET, or both. If lumbar puncture is contraindicated, PET investigations are an alternative.
- In clinical practice, plasma biomarkers for amyloid β and tau pathology are not currently recommended. Although promising, plasma biomarkers require further standardization and validation before they can be broadly regarded as secure evidence of Alzheimer’s disease pathology (amyloid-positive and tau-positive).71,105
- In clinical practice, the investigation of pathophysiological biomarkers in cognitively unimpaired individuals is not recommended, given the current inability to predict reliable clinical trajectories of people who are asymptomatic with biomarker positive status (amyloid-positive and tau-positive). In the future, if therapies or prevention programmes show substantial efficacy in delaying onset of disease, that will probably change the need for biomarker investigations in these individuals, although the problem of the prediction of clinical trajectories in cognitively unimpaired biomarker-positive individuals will still remain.
- Physicians are recommended to evaluate the added-value of biomarker investigation for each symptomatic patient objectively, according to the clinical situation (age, risk of comorbidity, complexity of the phenotype), the life context, the wishes of the patient to know the most likely diagnosis, the possibility of participation in a disease-modifying trial, and the appreciation of how this information will change the management of the patient. Biomarker investigations can also be limited by the availability, cost, and health-care payment coverage of biomarkers across countries, centres, and clinical situations” (Dubois et al., 2021).
Table 2 (Dubois et al., 2021):


United States Preventive Services Task Force (USPSTF)
In 2020, the USPSTF published a recommendation stating that “current evidence is insufficient to assess the balance of benefits and harms of screening for cognitive impairment in older adults” (Owens et al., 2020).
European Federation of Neurological Societies (EFNS)
The EFNS published updated guidelines in 2012 for the diagnosis and management of disorders associated with dementia. These guidelines state that “Routine CSF analysis may help to rule out or rule in certain infectious causes (Good Practice Point). CSF abeta 1‐42/tau/p‐tau assessment helps to differentiate AD (Level B). Assessment of CSF total tau and 14‐3‐3 protein is recommended in rapidly progressive dementia when sCJD is suspected (Good Practice Point)” (Sorbi et al., 2012).
Canadian Consensus Conference on the Diagnosis and Treatment of Dementia (CCCDTD)
In 2020, the CCCDTD released recommendations on the diagnosis and treatment of dementia. The guidelines state that “CSF analysis is not recommended routinely, but it can be considered in dementia patients with diagnostic uncertainty and onset at an early age (< 65) to rule out Alzheimer’s disease (AD) pathophysiology.” The guidelines also state that “CSF analysis can also be considered in dementia patients with diagnostic uncertainty and predominance of language, visuospatial, dysexecutive, or behavioral features to rule out AD pathophysiology” (Ismail et al., 2020).
National Institute for Health and Care Excellence (NICE)
In 2018, NICE released guidelines on assessment, management, and support for people living with dementia. With regards to further testing, NICE states to only consider them if “it would help diagnosed a dementia subtype and knowing more about the dementia subtype would change management.”
The NICE recommendations for further tests for Alzheimer’s disease are delineated below:
- “If the diagnosis is uncertain … and Alzheimer’s disease is suspected, consider either:
- FDG-PET (fluorodeoxyglucose-positron emission tomography-CT), or perfusion SPECT (single-photon emission CT) if FDG-PET is unavailable OR
- Examining cerebrospinal fluid for:
- Either total tau or total tau and phosphorylated-tau 181 and
- Either amyloid beta 1-42 or amyloid beta 1-42 and amyloid beta 1-40.
- If a diagnosis cannot be made after one of these tests, consider using the other one.
- Be aware that the older a person is, the more likely they are to get a false positive with cerebrospinal fluid examination.
- Do not rule out Alzheimer’s disease based solely on the results of CT or MRI scans.
- Do not use Apolipoprotein E genotyping or electroencephalography to diagnose Alzheimer’s disease.
- Be aware that young-onset Alzheimer’s disease has a genetic cause in some people” (NICE, 2018).
References:
- AAN. (2021). PRACTICE GUIDELINE UPDATE SUMMARY: MILD COGNITIVE IMPAIRMENT. https://www.aan.com/Guidelines/home/GuidelineDetail/881
- Albert, M. S., DeKosky, S. T., Dickson, D., Dubois, B., Feldman, H. H., Fox, N. C., Gamst, A., Holtzman, D. M., Jagust, W. J., Petersen, R. C., Snyder, P. J., Carrillo, M. C., Thies, B., & Phelps, C. H. (2011). The diagnosis of mild cognitive impairment due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement, 7(3), 270-279. https://doi.org/10.1016/j.jalz.2011.03.008
- Alzheimer's Association. (2024). Lecanemab Approved for Treatment of Early Alzheimer's Disease. https://www.alz.org/alzheimers-dementia/treatments/lecanemab-leqembi
- Arranz, A. M., & De Strooper, B. (2019). The role of astroglia in Alzheimer's disease: pathophysiology and clinical implications. Lancet Neurol, 18(4), 406-414. https://doi.org/10.1016/s1474-4422(18)30490-3
- Bennett, D. A., Wilson, R. S., Schneider, J. A., Evans, D. A., Beckett, L. A., Aggarwal, N. T., Barnes, L. L., Fox, J. H., & Bach, J. (2002). Natural history of mild cognitive impairment in older persons. Neurology, 59(2), 198-205.
- Blennow, K., Zetterberg, H., & Fagan, A. M. (2012). Fluid biomarkers in Alzheimer disease. Cold Spring Harb Perspect Med, 2(9), a006221. https://doi.org/10.1101/cshperspect.a006221
- BusinessWire. (2023, July 6). Quanterix Launches LucentAD Biomarker Blood Test to Aid Physician Diagnosis of Alzheimer’s Disease in Patients. https://www.businesswire.com/news/home/20230706742971/en/
- C2N. (2019). C2N Diagnostics Receives Breakthrough Device Designation from U.S. FDA for Blood Test to Screen for Alzheimer’s Disease Risk. https://c2n.com/news-releases/2019/01/29/2019-1-24-c2n-diagnostics-receives-breakthrough-device-designation-from-us-fda-for-blood-test-to-screen-for-alzheimers-disease-risk
- Chen, Y. R., Liang, C. S., Chu, H., Voss, J., Kang, X. L., O'Connell, G., Jen, H. J., Liu, D., Shen Hsiao, S. T., & Chou, K. R. (2021). Diagnostic accuracy of blood biomarkers for Alzheimer's disease and amnestic mild cognitive impairment: A meta-analysis. Ageing Res Rev, 71, 101446. https://doi.org/10.1016/j.arr.2021.101446
- CND Life Sciences. SYN-ONE TEST®. https://cndlifesciences.com/syn-one-test/
- Cordell, C. B., Borson, S., Boustani, M., Chodosh, J., Reuben, D., Verghese, J., Thies, W., & Fried, L. B. (2013). Alzheimer's Association recommendations for operationalizing the detection of cognitive impairment during the Medicare Annual Wellness Visit in a primary care setting. Alzheimers Dement, 9(2), 141-150. https://doi.org/10.1016/j.jalz.2012.09.011
- Dage, J. L., Wennberg, A. M. V., Airey, D. C., Hagen, C. E., Knopman, D. S., Machulda, M. M., Roberts, R. O., Jack, C. R., Jr., Petersen, R. C., & Mielke, M. M. (2016). Levels of tau protein in plasma are associated with neurodegeneration and cognitive function in a population-based elderly cohort. Alzheimers Dement, 12(12), 1226-1234. https://doi.org/10.1016/j.jalz.2016.06.001
- Donadio, V., Doppler, K., Incensi, A., Kuzkina, A., Janzen, A., Mayer, G., Volkmann, J., Rizzo, G., Antelmi, E., Plazzi, G., Sommer, C., Liguori, R., & Oertel, W. H. (2019). Abnormal alpha-synuclein deposits in skin nerves: intra- and inter-laboratory reproducibility. Eur J Neurol, 26(10), 1245-1251. https://doi.org/10.1111/ene.13939
- Dubois, B., Feldman, H. H., Jacova, C., Hampel, H., Molinuevo, J. L., Blennow, K., DeKosky, S. T., Gauthier, S., Selkoe, D., Bateman, R., Cappa, S., Crutch, S., Engelborghs, S., Frisoni, G. B., Fox, N. C., Galasko, D., Habert, M. O., Jicha, G. A., Nordberg, A., . . . Cummings, J. L. (2014). Advancing research diagnostic criteria for Alzheimer's disease: the IWG-2 criteria. Lancet Neurol, 13(6), 614-629. https://doi.org/10.1016/s1474-4422(14)70090-0
- Dubois, B., Villain, N., Frisoni, G. B., Rabinovici, G. D., Sabbagh, M., Cappa, S., Bejanin, A., Bombois, S., Epelbaum, S., Teichmann, M., Habert, M. O., Nordberg, A., Blennow, K., Galasko, D., Stern, Y., Rowe, C. C., Salloway, S., Schneider, L. S., Cummings, J. L., & Feldman, H. H. (2021). Clinical diagnosis of Alzheimer's disease: recommendations of the International Working Group. Lancet Neurol, 20(6), 484-496. https://doi.org/10.1016/s1474-4422(21)00066-1
- Farrer, L. A., Cupples, L. A., Haines, J. L., Hyman, B., Kukull, W. A., Mayeux, R., Myers, R. H., Pericak-Vance, M. A., Risch, N., & van Duijn, C. M. (1997). Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis. APOE and Alzheimer Disease Meta Analysis Consortium. Jama, 278(16), 1349-1356. http://dx.doi.org/
- FDA. (2022, May 4). FDA Permits Marketing for New Test to Improve Diagnosis of Alzheimer’s Disease. https://www.prnewswire.com/news-releases/fda-permits-marketing-for-new-test-to-improve-diagnosis-of-alzheimers-disease-301540093.html
- FDA. (2024, 01/29/2018). Early Alzheimer's Disease: Developing Drugs for Treatment. U.S. Food and Drug Administration Center for Drug Evaluation and Research Center for Biologics Evaluation and Research. Retrieved 08/31/2018 from https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM596728.pdf
- Fossati, S., Ramos Cejudo, J., Debure, L., Pirraglia, E., Sone, J. Y., Li, Y., Chen, J., Butler, T., Zetterberg, H., Blennow, K., & de Leon, M. J. (2019). Plasma tau complements CSF tau and P-tau in the diagnosis of Alzheimer's disease. Alzheimers Dement (Amst), 11, 483-492. https://doi.org/10.1016/j.dadm.2019.05.001
- François, M., Bull, C. F., Fenech, M. F., & Leifert, W. R. (2019). Current State of Saliva Biomarkers for Aging and Alzheimer's Disease. Curr Alzheimer Res, 16(1), 56-66. https://doi.org/10.2174/1567205015666181022094924
- Fujirebio. (2022). Lumipulse® G β-Amyloid Ratio (1-42/1-40). https://www.fujirebio.com/en-us/products-solutions/lumipulse-g-beta-amyloid-ratio-142-140
- Gatz, M., Reynolds, C. A., Fratiglioni, L., Johansson, B., Mortimer, J. A., Berg, S., Fiske, A., & Pedersen, N. L. (2006). Role of genes and environments for explaining Alzheimer disease. Arch Gen Psychiatry, 63(2), 168-174. https://doi.org/10.1001/archpsyc.63.2.168
- GBSC. (2024). Biomarker Consortium. Global Biomarker Standardization Consortium (GBSC); Alzheimer's Association. https://www.alz.org/research/for_researchers/partnerships/biomarker_consortium
- Goldman, J. S., Hahn, S. E., Catania, J. W., LaRusse-Eckert, S., Butson, M. B., Rumbaugh, M., Strecker, M. N., Roberts, J. S., Burke, W., Mayeux, R., & Bird, T. (2011). Genetic counseling and testing for Alzheimer disease: Joint practice guidelines of the American College of Medical Genetics and the National Society of Genetic Counselors. Genet Med, 13(6), 597-605. https://doi.org/10.1097/GIM.0b013e31821d69b8
- Gottlieb, S. (2018, 02/15/2018). Statement from FDA Commissioner Scott Gottlieb, M.D. on advancing the development of novel treatments for neurological conditions; part of broader effort on modernizing FDA’s new drug review programs. U.S. Food and Drug Administration. Retrieved 08/31/2018 from https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm596897.htm
- Hansson, O., Edelmayer, R. M., Boxer, A. L., Carrillo, M. C., Mielke, M. M., Rabinovici, G. D., Salloway, S., Sperling, R., Zetterberg, H., & Teunissen, C. E. (2022). The Alzheimer's Association appropriate use recommendations for blood biomarkers in Alzheimer's disease. Alzheimers Dement, 18(12), 2669-2686. https://doi.org/10.1002/alz.12756
- Hansson, O., Lehmann, S., Otto, M., Zetterberg, H., & Lewczuk, P. (2019). Advantages and disadvantages of the use of the CSF Amyloid β (Aβ) 42/40 ratio in the diagnosis of Alzheimer's Disease. Alzheimers Res Ther, 11(1), 34. https://doi.org/10.1186/s13195-019-0485-0
- Herukka, S. K., Simonsen, A. H., Andreasen, N., Baldeiras, I., Bjerke, M., Blennow, K., Engelborghs, S., Frisoni, G. B., Gabryelewicz, T., Galluzzi, S., Handels, R., Kramberger, M. G., Kulczyńska, A., Molinuevo, J. L., Mroczko, B., Nordberg, A., Oliveira, C. R., Otto, M., Rinne, J. O., . . . Waldemar, G. (2017). Recommendations for cerebrospinal fluid Alzheimer's disease biomarkers in the diagnostic evaluation of mild cognitive impairment. Alzheimers Dement, 13(3), 285-295. https://doi.org/10.1016/j.jalz.2016.09.009
- Huan, T., Tran, T., Zheng, J., Sapkota, S., MacDonald, S. W., Camicioli, R., Dixon, R. A., & Li, L. (2018). Metabolomics Analyses of Saliva Detect Novel Biomarkers of Alzheimer's Disease. J Alzheimers Dis, 65(4), 1401-1416. https://doi.org/10.3233/jad-180711
- Ismail, Z., Black, S. E., Camicioli, R., Chertkow, H., Herrmann, N., Laforce, R., Jr., Montero-Odasso, M., Rockwood, K., Rosa-Neto, P., Seitz, D., Sivananthan, S., Smith, E. E., Soucy, J. P., Vedel, I., & Gauthier, S. (2020). Recommendations of the 5th Canadian Consensus Conference on the diagnosis and treatment of dementia. Alzheimers Dement, 16(8), 1182-1195. https://doi.org/10.1002/alz.12105
- Jack, C. R., Jr., Bennett, D. A., Blennow, K., Carrillo, M. C., Dunn, B., Haeberlein, S. B., Holtzman, D. M., Jagust, W., Jessen, F., Karlawish, J., Liu, E., Molinuevo, J. L., Montine, T., Phelps, C., Rankin, K. P., Rowe, C. C., Scheltens, P., Siemers, E., Snyder, H. M., & Sperling, R. (2018). NIA-AA Research Framework: Toward a biological definition of Alzheimer's disease. Alzheimers Dement, 14(4), 535-562. https://doi.org/10.1016/j.jalz.2018.02.018
- Jiang, L., Dong, H., Cao, H., Ji, X., Luan, S., & Liu, J. (2019). Exosomes in Pathogenesis, Diagnosis, and Treatment of Alzheimer's Disease. Med Sci Monit, 25, 3329-3335. https://doi.org/10.12659/msm.914027
- Karch, C. M., Cruchaga, C., & Goate, A. M. (2014). Alzheimer's disease genetics: from the bench to the clinic. Neuron, 83(1), 11-26. https://doi.org/10.1016/j.neuron.2014.05.041
- Kim, J. Y., Illigens, B. M., McCormick, M. P., Wang, N., & Gibbons, C. H. (2019). Alpha-Synuclein in Skin Nerve Fibers as a Biomarker for Alpha-Synucleinopathies. J Clin Neurol, 15(2), 135-142. https://doi.org/10.3988/jcn.2019.15.2.135
- Kim, K., Kim, M.-J., Kim, D. W., Kim, S. Y., Park, S., & Park, C. B. (2020). Clinically accurate diagnosis of Alzheimer’s disease via multiplexed sensing of core biomarkers in human plasma. Nature Communications, 11(1), 119. https://doi.org/10.1038/s41467-019-13901-z
- Knopman, D. S., DeKosky, S. T., Cummings, J. L., Chui, H., Corey-Bloom, J., Relkin, N., Small, G. W., Miller, B., & Stevens, J. C. (2001). Practice parameter: diagnosis of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology, 56(9), 1143-1153. https://doi.org/10.1212/wnl.56.9.1143
- Kramarow, E., & Tejada-Vera, B. (2019). Adjusted Death Rates* from Dementia,† by Sex, Race, and Hispanic Origin — National Vital Statistics System, United States, 2017. MMWR Morb Mortal Wkly Rep, 68, 670. https://www.cdc.gov/mmwr/volumes/68/wr/mm6830a6.htm?s_cid=mm6830a6_w
- Levine, T. D., Bellaire, B., Gibbons, C., & Freeman, R. (2021). Cutaneous alpha-synuclein deposition in postural tachycardia patients. Ann Clin Transl Neurol, 8(4), 908-917. https://doi.org/10.1002/acn3.51347
- Lewczuk, P., Matzen, A., Blennow, K., Parnetti, L., Molinuevo, J. L., Eusebi, P., Kornhuber, J., Morris, J. C., & Fagan, A. M. (2017). Cerebrospinal Fluid Abeta42/40 Corresponds Better than Abeta42 to Amyloid PET in Alzheimer's Disease. J Alzheimers Dis, 55(2), 813-822. https://doi.org/10.3233/jad-160722
- Lewczuk, P., Riederer, P., O'Bryant, S. E., Verbeek, M. M., Dubois, B., Visser, P. J., Jellinger, K. A., Engelborghs, S., Ramirez, A., Parnetti, L., Jack, C. R., Jr., Teunissen, C. E., Hampel, H., Lleó, A., Jessen, F., Glodzik, L., de Leon, M. J., Fagan, A. M., Molinuevo, J. L., . . . Kornhuber, J. (2018). Cerebrospinal fluid and blood biomarkers for neurodegenerative dementias: An update of the Consensus of the Task Force on Biological Markers in Psychiatry of the World Federation of Societies of Biological Psychiatry. World J Biol Psychiatry, 19(4), 244-328. https://doi.org/10.1080/15622975.2017.1375556
- Liu, Y., Wei, M., Yue, K., Hu, M., Li, S., Men, L., Pi, Z., Liu, Z., & Liu, Z. (2018). Study on Urine Metabolic Profile of Abeta25-35-Induced Alzheimer's Disease Using UHPLC-Q-TOF-MS. Neuroscience, 394, 30-43. https://doi.org/10.1016/j.neuroscience.2018.10.001
- Lonneborg, A. (2008). Biomarkers for Alzheimer disease in cerebrospinal fluid, urine, and blood. Mol Diagn Ther, 12(5), 307-320.
- Mattsson, N., Andreasson, U., Persson, S., Arai, H., Batish, S. D., Bernardini, S., Bocchio-Chiavetto, L., Blankenstein, M. A., Carrillo, M. C., Chalbot, S., Coart, E., Chiasserini, D., Cutler, N., Dahlfors, G., Duller, S., Fagan, A. M., Forlenza, O., Frisoni, G. B., Galasko, D., . . . Blennow, K. (2011). The Alzheimer’s Association external quality control program for cerebrospinal fluid biomarkers. Alzheimers Dement, 7(4), 386-395 e386. https://doi.org/10.1016/j.jalz.2011.05.2243
- McKeith, I. G., Boeve, B. F., Dickson, D. W., Halliday, G., Taylor, J. P., Weintraub, D., Aarsland, D., Galvin, J., Attems, J., Ballard, C. G., Bayston, A., Beach, T. G., Blanc, F., Bohnen, N., Bonanni, L., Bras, J., Brundin, P., Burn, D., Chen-Plotkin, A., . . . Kosaka, K. (2017). Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium. Neurology, 89(1), 88-100. https://doi.org/10.1212/wnl.0000000000004058
- McKhann, G. M., Knopman, D. S., Chertkow, H., Hyman, B. T., Jack, C. R., Kawas, C. H., Klunk, W. E., Koroshetz, W. J., Manly, J. J., Mayeux, R., Mohs, R. C., Morris, J. C., Rossor, M. N., Scheltens, P., Carrillo, M. C., Thies, B., Weintraub, S., & Phelps, C. H. (2011). The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement, 7(3), 263-269. https://doi.org/10.1016/j.jalz.2011.03.005
- Morris, J. C., Blennow, K., Froelich, L., Nordberg, A., Soininen, H., Waldemar, G., Wahlund, L. O., & Dubois, B. (2014). Harmonized diagnostic criteria for Alzheimer's disease: recommendations. J Intern Med, 275(3), 204-213. https://doi.org/10.1111/joim.12199
- NICE. (2018, June 20). Dementia: assessment, management and support for people living with dementia and their carers. https://www.nice.org.uk/guidance/ng97/chapter/Recommendations
- Nojima, H., Ito, S., Kushida, A., Abe, A., Motsuchi, W., Verbel, D., Vandijck, M., Jannes, G., Vandenbroucke, I., & Aoyagi, K. (2022). Clinical utility of cerebrospinal fluid biomarkers measured by LUMIPULSE(®) system. Ann Clin Transl Neurol, 9(12), 1898-1909. https://doi.org/10.1002/acn3.51681
- Owens, D. K., Davidson, K. W., Krist, A. H., Barry, M. J., Cabana, M., Caughey, A. B., Doubeni, C. A., Epling, J. W., Jr., Kubik, M., Landefeld, C. S., Mangione, C. M., Pbert, L., Silverstein, M., Simon, M. A., Tseng, C. W., & Wong, J. B. (2020). Screening for Cognitive Impairment in Older Adults: US Preventive Services Task Force Recommendation Statement. Jama, 323(8), 757-763. https://doi.org/10.1001/jama.2020.0435
- Palmqvist, S., Janelidze, S., Stomrud, E., Zetterberg, H., Karl, J., Zink, K., Bittner, T., Mattsson, N., Eichenlaub, U., Blennow, K., & Hansson, O. (2019). Performance of Fully Automated Plasma Assays as Screening Tests for Alzheimer Disease-Related β-Amyloid Status. JAMA Neurol, 76(9), 1060-1069. https://doi.org/10.1001/jamaneurol.2019.1632
- Patel, S., Shah, R. J., Coleman, P., & Sabbagh, M. (2011). Potential peripheral biomarkers for the diagnosis of Alzheimer's disease. Int J Alzheimers Dis, 2011, 572495. https://doi.org/10.4061/2011/572495
- Peterson, R. C. (2024, July 30). Mild cognitive impairment: Prognosis and treatment. https://www.uptodate.com/contents/mild-cognitive-impairment-prognosis-and-treatment
- Qu, Y., Ma, Y. H., Huang, Y. Y., Ou, Y. N., Shen, X. N., Chen, S. D., Dong, Q., Tan, L., & Yu, J. T. (2021). Blood biomarkers for the diagnosis of amnestic mild cognitive impairment and Alzheimer's disease: A systematic review and meta-analysis. Neurosci Biobehav Rev, 128, 479-486. https://doi.org/10.1016/j.neubiorev.2021.07.007
- Roche. (2022, December 8). Roche Alzheimer’s disease Cerebrospinal Fluid (CSF) assays receive FDA clearance, supporting more accurate and timely diagnosis. https://www.globenewswire.com/news-release/2022/12/08/2569879/0/en/Roche-Alzheimer-s-disease-Cerebrospinal-Fluid-CSF-assays-receive-FDA-clearance-supporting-more-accurate-and-timely-diagnosis.html
- Roche. (2023, June 27). Roche receives FDA clearance for additional Alzheimer’s disease Cerebrospinal Fluid (CSF) assays, supporting timely diagnosis and treatment decision-making. https://diagnostics.roche.com/us/en/news-listing/2023/roche-fda-clearance-additional-alzheimers-disease-cerebrospinal-fluid-ttau.html
- Sala Frigerio, C., & De Strooper, B. (2016). Alzheimer's Disease Mechanisms and Emerging Roads to Novel Therapeutics. Annual Review of Neuroscience, 39(1), 57-79. https://doi.org/10.1146/annurev-neuro-070815-014015
- Schaffer, C., Sarad, N., DeCrumpe, A., Goswami, D., Herrmann, S., Morales, J., Patel, P., & Osborne, J. (2015). Biomarkers in the Diagnosis and Prognosis of Alzheimer's Disease. J Lab Autom, 20(5), 589-600. https://doi.org/10.1177/2211068214559979
- Shaw, L. M., Arias, J., Blennow, K., Galasko, D., Molinuevo, J. L., Salloway, S., Schindler, S., Carrillo, M. C., Hendrix, J. A., Ross, A., Illes, J., Ramus, C., & Fifer, S. (2018). Appropriate use criteria for lumbar puncture and cerebrospinal fluid testing in the diagnosis of Alzheimer's disease. Alzheimers Dement, 14(11), 1505-1521. https://doi.org/10.1016/j.jalz.2018.07.220
- Shea, Y. F., Chu, L. W., Chan, A. O., Ha, J., Li, Y., & Song, Y. Q. (2016). A systematic review of familial Alzheimer's disease: Differences in presentation of clinical features among three mutated genes and potential ethnic differences. J Formos Med Assoc, 115(2), 67-75. https://doi.org/10.1016/j.jfma.2015.08.004
- Shen, X. N., Niu, L. D., Wang, Y. J., Cao, X. P., Liu, Q., Tan, L., Zhang, C., & Yu, J. T. (2019). Inflammatory markers in Alzheimer's disease and mild cognitive impairment: a meta-analysis and systematic review of 170 studies. J Neurol Neurosurg Psychiatry, 90(5), 590-598. https://doi.org/10.1136/jnnp-2018-319148
- Simonsen, A. H., Herukka, S. K., Andreasen, N., Baldeiras, I., Bjerke, M., Blennow, K., Engelborghs, S., Frisoni, G. B., Gabryelewicz, T., Galluzzi, S., Handels, R., Kramberger, M. G., Kulczyńska, A., Molinuevo, J. L., Mroczko, B., Nordberg, A., Oliveira, C. R., Otto, M., Rinne, J. O., . . . Waldemar, G. (2017). Recommendations for CSF AD biomarkers in the diagnostic evaluation of dementia. Alzheimers Dement, 13(3), 274-284. https://doi.org/10.1016/j.jalz.2016.09.008
- Simrén, J., Leuzy, A., Karikari, T. K., Hye, A., Benedet, A. L., Lantero-Rodriguez, J., Mattsson-Carlgren, N., Schöll, M., Mecocci, P., Vellas, B., Tsolaki, M., Kloszewska, I., Soininen, H., Lovestone, S., Aarsland, D., consortium, f. t. A., Hansson, O., Rosa-Neto, P., Westman, E., . . . Ashton, N. J. (2021). The diagnostic and prognostic capabilities of plasma biomarkers in Alzheimer's disease. Alzheimer's & Dementia, 17(7), 1145-1156. https://doi.org/https://doi.org/10.1002/alz.12283
- Sorbi, S., Hort, J., Erkinjuntti, T., Fladby, T., Gainotti, G., Gurvit, H., Nacmias, B., Pasquier, F., Popescu, B. O., Rektorova, I., Religa, D., Rusina, R., Rossor, M., Schmidt, R., Stefanova, E., Warren, J. D., & Scheltens, P. (2012). EFNS-ENS Guidelines on the diagnosis and management of disorders associated with dementia. Eur J Neurol, 19(9), 1159-1179. https://doi.org/10.1111/j.1468-1331.2012.03784.x
- Talwar, P., Sinha, J., Grover, S., Agarwal, R., Kushwaha, S., Srivastava, M. V., & Kukreti, R. (2016). Meta-analysis of apolipoprotein E levels in the cerebrospinal fluid of patients with Alzheimer's disease. J Neurol Sci, 360, 179-187. https://doi.org/10.1016/j.jns.2015.12.004
- Tatebe, H., Kasai, T., Ohmichi, T., Kishi, Y., Kakeya, T., Waragai, M., Kondo, M., Allsop, D., & Tokuda, T. (2017). Quantification of plasma phosphorylated tau to use as a biomarker for brain Alzheimer pathology: pilot case-control studies including patients with Alzheimer's disease and down syndrome. Mol Neurodegener, 12(1), 63. https://doi.org/10.1186/s13024-017-0206-8
- Van Cauwenberghe, C., Van Broeckhoven, C., & Sleegers, K. (2016). The genetic landscape of Alzheimer disease: clinical implications and perspectives. Genet Med, 18(5), 421-430. https://doi.org/10.1038/gim.2015.117
- Wang, H., Stewart, T., Toledo, J. B., Ginghina, C., Tang, L., Atik, A., Aro, P., Shaw, L. M., Trojanowski, J. Q., Galasko, D. R., Edland, S., Jensen, P. H., Shi, M., & Zhang, J. (2018). A Longitudinal Study of Total and Phosphorylated alpha-Synuclein with Other Biomarkers in Cerebrospinal Fluid of Alzheimer's Disease and Mild Cognitive Impairment. J Alzheimers Dis, 61(4), 1541-1553. https://doi.org/10.3233/jad-171013
- Wang, S., Kojima, K., Mobley, J. A., & West, A. B. (2019). Proteomic analysis of urinary extracellular vesicles reveal biomarkers for neurologic disease. EBioMedicine, 45, 351-361. https://doi.org/10.1016/j.ebiom.2019.06.021
- Waqar, S., Khan, H., Zulfiqar, S. K., & Ahmad, A. (2023). Skin Biopsy as a Diagnostic Tool for Synucleinopathies. Cureus, 15(10), e47179. https://doi.org/10.7759/cureus.47179
- WHO. (2023, 03/15/2023). Dementia. https://www.who.int/news-room/fact-sheets/detail/dementia
- Wolk, D. A., & Dickerson, B. C. (2024, October 8). Clinical features and diagnosis of Alzheimer disease. https://www.uptodate.com/contents/clinical-features-and-diagnosis-of-alzheimer-disease
- Yoong, S. Q., Lu, J., Xing, H., Gyanwali, B., Tan, Y. Q., & Wu, X. V. (2021). The prognostic utility of CSF neurogranin in predicting future cognitive decline in the Alzheimer's disease continuum: A systematic review and meta-analysis with narrative synthesis. Ageing Res Rev, 72, 101491. https://doi.org/10.1016/j.arr.2021.101491
- Zhang, J., Zhang, C. H., Li, R. J., Lin, X. L., Chen, Y. D., Gao, H. Q., & Shi, S. L. (2014). Accuracy of urinary AD7c-NTP for diagnosing Alzheimer's disease: a systematic review and meta-analysis. J Alzheimers Dis, 40(1), 153-159. https://doi.org/10.3233/jad-131445
Coding Section
| Codes | Number | Description |
| CPT | 82233 (effective 01/01/2025) | Beta-amyloid; 1-40 (Abeta 40) |
| 82234 (effective 01/01/2025) | Beta-amyloid; 1-42 (Abeta 42) | |
| 83884 (effective 01/01/2025) | Neurofilament light chain (NfL) | |
| 84393 (effective 01/01/2025) | Tau, phosphorylated (eg, pTau 181, pTau 217), each | |
| 84394 (effective 01/01/2025) | Tau, total (tTau) | |
| 83520 | Immunoassay for analyte other than infectious agent antibody or infectious agent antigen; quantitative, not otherwise specified | |
| 0206U | Neurology (Alzheimer disease); cell aggregation using morphometric imaging and protein kinase C-epsilon (PKCe) concentration in response to amylospheroid treatment by ELISA, cultured skin fibroblasts, each reported as positive or negative for Alzheimer disease | |
| 0207U | Quantitative imaging of phosphorylated ERK1 and ERK2 in response to bradykinin treatment by in situ immunofluorescence, using cultured skin fibroblasts, reported as a probability index for Alzheimer disease (List separately in addition to code for primary procedure) | |
| 0289U | Neurology (Alzheimer disease), mRNA, gene expression profiling by RNA sequencing of 24 genes, whole blood, algorithm reported as predictive risk score Proprietary test: MindX Blood Test™ — Memory/Alzheimer's Lab/Manufacturer: MindX Sciences™ Laboratory/MindX Sciences™ Inc |
|
| 0346U | Beta amyloid, Aβ40 and Aβ42 by liquid chromatography with tandem mass spectrometry (LC-MS/MS), ratio, plasma Protietary test: QUEST AD-Detect™, Beta-Amyloid 42/40 Ratio, Plasma Lab/Manufacturer: Quest Diagnostics |
|
| 0358U | Neurology (mild cognitive impairment), analysis of β-amyloid 1-42 and 1-40, chemiluminescence enzyme immunoassay, cerebral spinal fluid, reported as positive, likely positive, or negative Proprietary test: Lumipulse® G β-Amyloid Ratio (1-42/1-40) Test Lab/Manufacturer: Fujirebio Diagnostics, Inc | |
| 0393U (effective 07/01/2025) | Neurology (eg, Parkinson disease, dementia with Lewy bodies), cerebrospinal fluid (CSF), detection of misfolded α-synuclein protein by seed amplification assay, qualitative | |
| 0412U (effective 10/01/2023) | Beta amyloid, Aβ42/40 ratio, immunoprecipitation with quantitation by liquid chromatography with tandem mass spectrometry (LC-MS/MS) and qualitative ApoE isoform-specific proteotyping, plasma combined with age, algorithm reported as presence or absence of brain amyloid pathology | |
| 0443U (effective 04/01/2024) | Neurofilament light chain (NfL), ultra-sensitive immunoassay, serum or cerebrospinal fluid | |
| 0445U (effective 04/01/2024) | β-amyloid (Abeta42) and phospho tau (181P) (pTau181), electrochemiluminescent immunoassay (ECLIA), cerebral spinal fluid, ratio reported as positive or negative for amyloid pathology | |
| 0459U | β-amyloid (Abeta42) and total tau (tTau), electrochemiluminescent immunoassay (ECLIA), cerebral spinal fluid, ratio reported as positive or negative for amyloid pathology Proprietary test: Elecsys® Total Tau CSF (tTau) and β-Amyloid (1-42) CSF II (Abeta 42) Ratio Lab/Manufacturer: Roche Diagnostics Operations, Inc (US owner/operator) |
|
| 0479U (effective 10/01/2024) | Tau, phosphorylated, pTau217 (effective on 10/01/2024) | |
| 0503U (effective 10/01/2024) | Neurology (Alzheimer disease), beta amyloid (Aβ40, Aβ42, Aβ42/40 ratio) and tau-protein (ptau217, np-tau217, ptau217/nptau217 ratio), blood, immunoprecipitation with quantitation by liquid chromatography with tandem mass spectrometry (LC-MS/MS), algorithm score reported as ikelihood of positive or negative for amyloid plaques | |
| 0547U (effective 04/01/2025) | Neurofilament light chain (NfL), chemiluminescent enzyme immunoassay, plasma, quantitative | |
| 0548U (effective 04/01/2025) | Glial fibrillary acidic protein (GFAP), chemiluminescent enzyme immunoassay, using plasma | |
| 0551U (effective 04/01/2025) | Tau, phosphorylated, pTau217, by single-molecule array (ultrasensitive digital protein detection), using plasma | |
| 0568U (effective 07/01/2025) | Neurology (dementia), beta amyloid (A?40, A?42, A?42/40 ratio), tau-protein phosphorylated at residue (eg, pTau217), neurofilament light chain (NfL), and glial fibrillary acidic protein (GFAP), by ultra-high sensitivity molecule array detection, plasma, algorithm reported as positive, intermediate, or negative for Alzheimer pathology | |
| HCPCS | ||
| ICD-10-CM (effective 10/01/15) | ||
| F03 | Unspecified dementia | |
| G30.0-G30.9 | Alzheimer disease code range | |
| G31.1 | Senile degeneration of brain, not elsewhere classified | |
| R41.0 | Disorientation, unspecified | |
| R41.81 | Age-related cognitive decline | |
| Z13.858 | Encounter for screening for other nervous system disorders | |
| ICD-10-PCS (effective 10/01/15) | Not applicable. ICD-10-PCS codes are only used for inpatient services. There are no ICD procedure codes for laboratory tests. | |
| Type of Service | Pathology/Laboratory | |
| Place of Service | Physician's Office |
Procedure and diagnosis codes on Medical Policy documents are included only as a general reference tool for each policy. They may not be all-inclusive.
This medical policy was developed through consideration of peer-reviewed medical literature generally recognized by the relevant medical community, U.S. FDA approval status, nationally accepted standards of medical practice and accepted standards of medical practice in this community, Blue Cross Blue Shield Association technology assessment program (TEC) and other nonaffiliated technology evaluation centers, reference to federal regulations, other plan medical policies and accredited national guidelines.
"Current Procedural Terminology © American Medical Association. All Rights Reserved"
History From 2024 Forward